The Conversation (0)
Sign Up

1st Step: Write the equation.
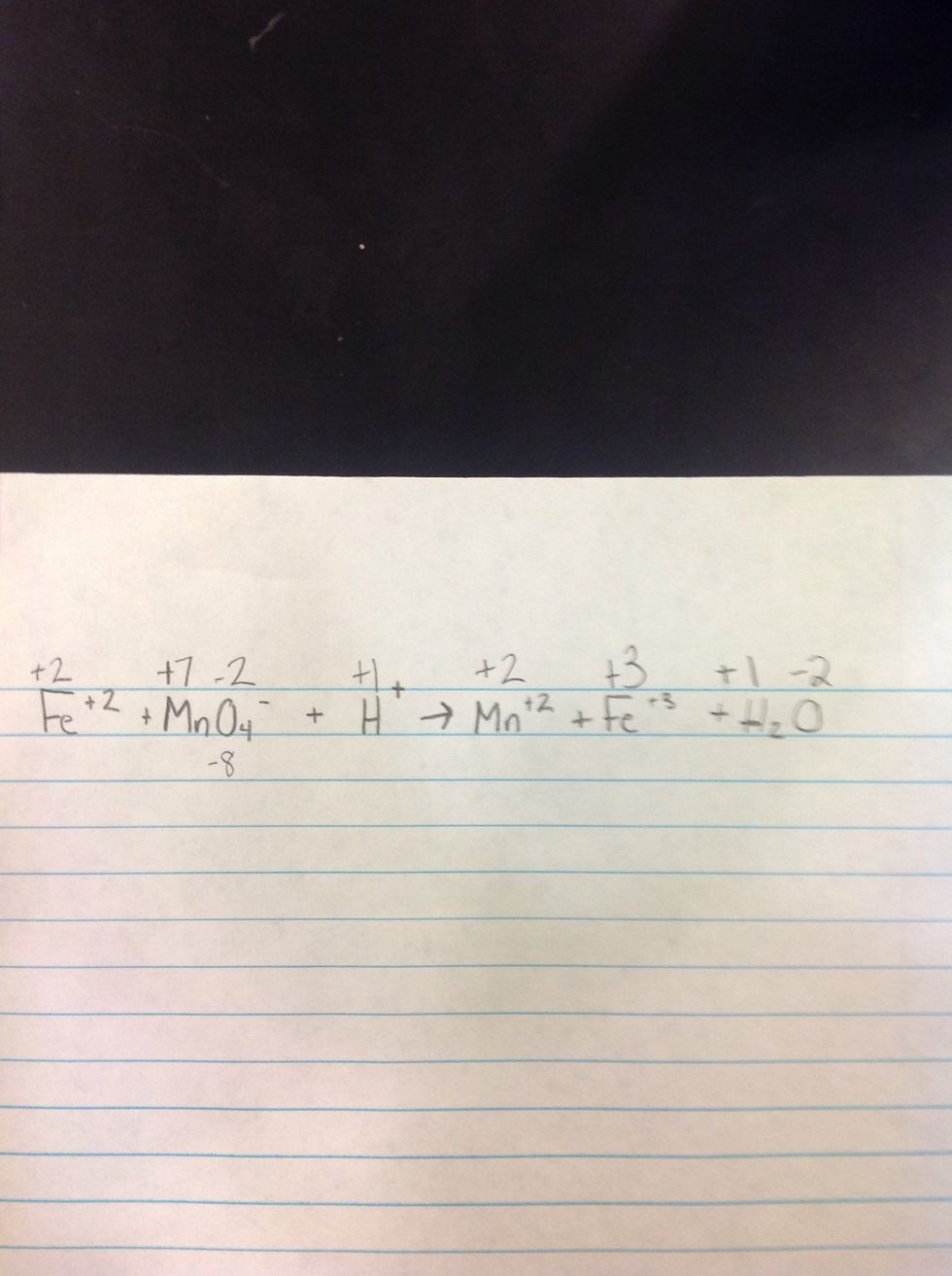
2nd Step: Determine the charges of each element.
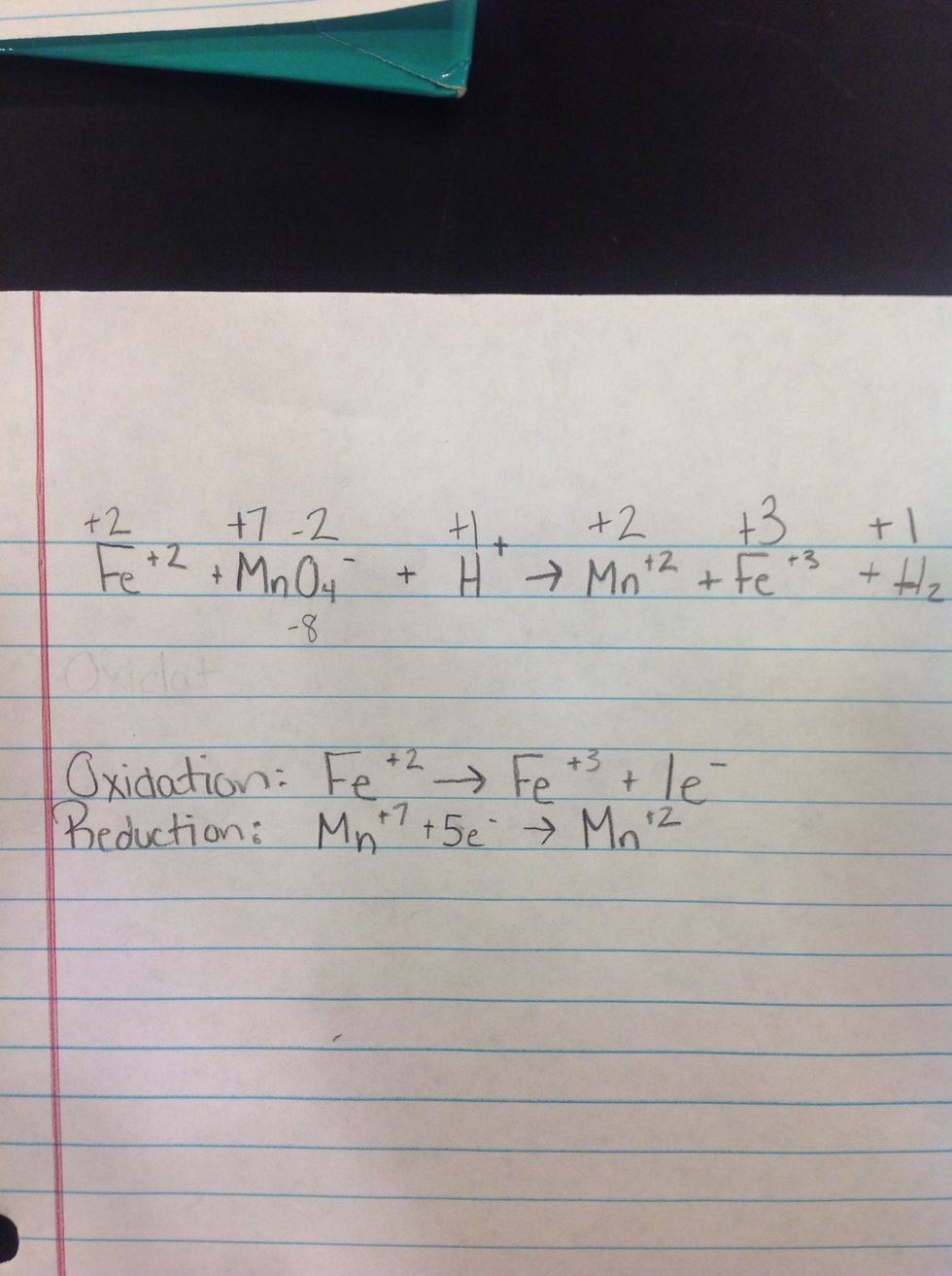
3rd Step: Determine which is oxidation and reduction and how many electrons they need. Electrons need to be the same in both reduction and oxidation.
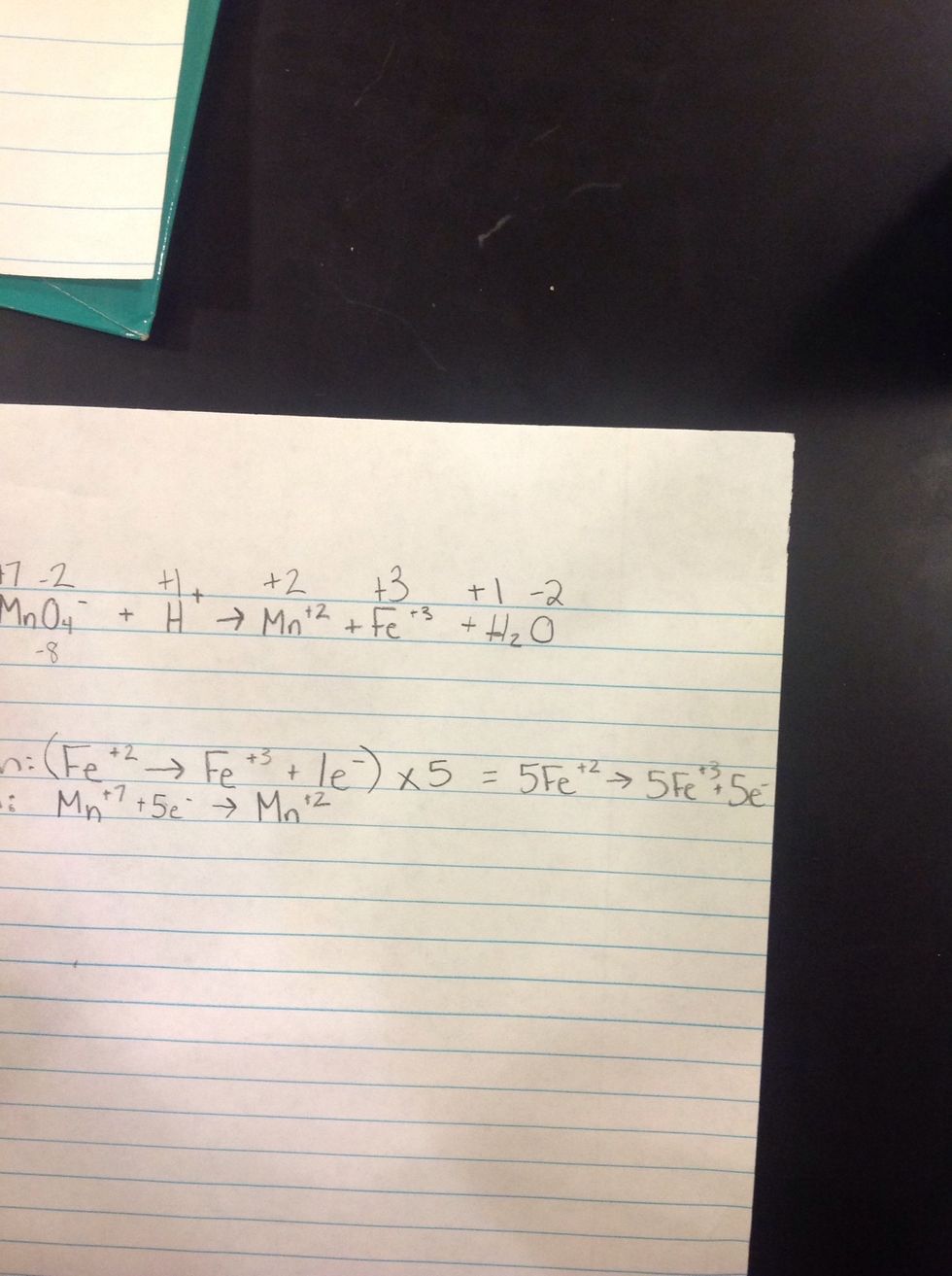
4th Step: Get both equations to have the same electrons and then rewrite the equation.
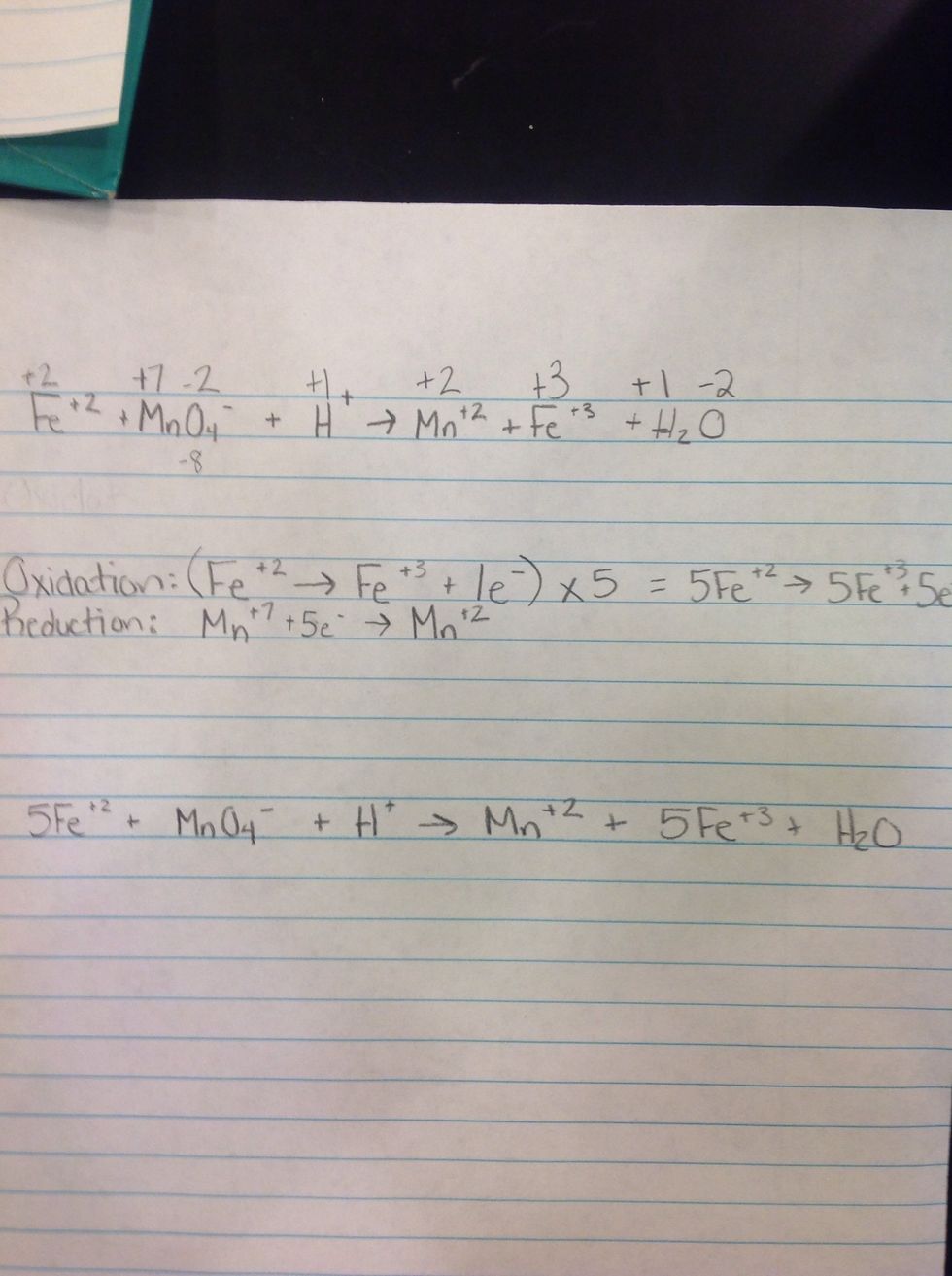
5th Step. Rewrite the whole entire equation using the rewritten half reactions.
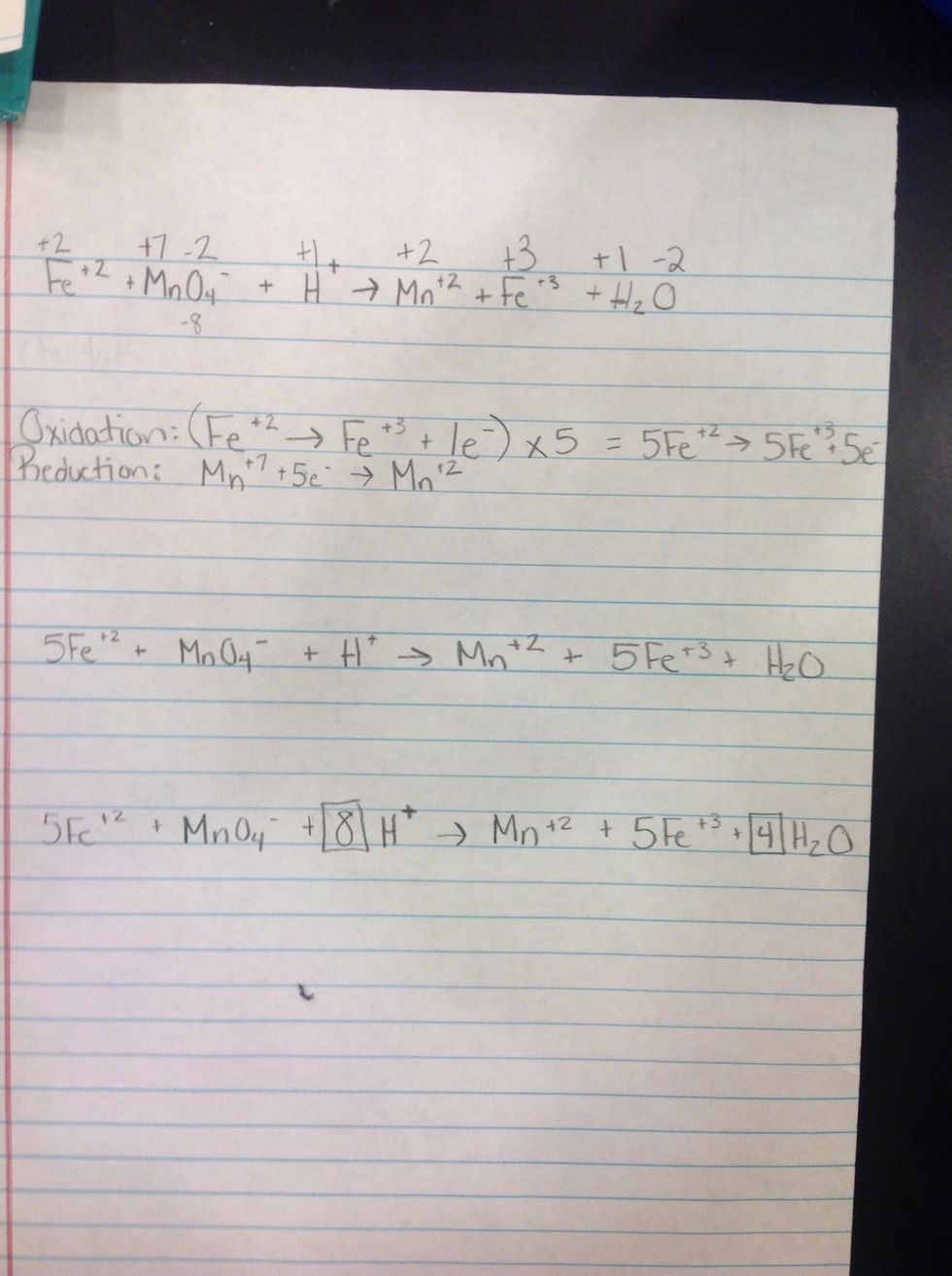
Last Step: Balance the whole entire equation.